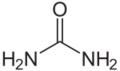
Urea
Jump to navigation
Jump to search
A major source of organic nitrogen.
Uses
Primary
- Industrial chemical
- Fertilizer (46-0-0)
Secondary
- Feedstock (through natural decomposition) to ammonia
- Feedstock for urea nitrate for cold packs
- Pharm: (ointment) Skin treatment
Natural occurrence
Hazards
Production
Extraction
from urine
Approximately 9.3g are expected in a liter of fresh human urine. Delay in processing allows bacteria to convert the urea to ammonia.
Pee foam
- Prussian blue
- DBX Labs
- Hyperspace pirate
simple evaporation
- Dry urine to solids
- Repeat
- Dissolve in ethanol
- filter
- discard residue
- evaporate alcohol
- gather crystals
- Until nothing but clear crystals remains
The crystals are urea
further separation
- Reactants
- Products
- Urea
- Mineral salts rich in Sodium chloride (roughly sea salt)
- Organic sludge rich in phosphate and proteins
- Equipment
- Liquid Dryer/Evaporater
- Gas bubbling equipment
- (Optional) Distillation equipment
- Process
- Boil 10L of urine down to 1L (containing ~93g, 6.6M of urea)
- Filter
- Residue contains organics, calcium and other phosphates, can be retained to produce phosphorus or burned and disposed
- Filtrate contains sodium chloride and urea
- Evaporate water by gentle heating (≤ 133°C)
- Residue is mostly sodium chloride and urea
- Wash residue with hot concentrated ethanol several times, combine washings
- NB: Urea dissolves easily, salt does not.
- IF (two layers form in the washings)
- Separate the aqueous layer and evaporate separately, producing mineral salts
- ENDIF
- Evaporate ethanol washings
- NB: It may be worth condensing the ethanol vapors
- The residue is urea. It should crystallize
via oxalic acid
- Precipitate from urine using oxalic acid, then extract again using calcium hydroxide. Detoxify the calcium oxalate by thermal decomposition.
- {{#Chem: CO(NH2)2 + H2(COO)2 = C3H6N2O5}}
- {{#Chem: C3H6N2O5 + Ca(OH)2 = CO(NH2)2 + Ca(COO)2 + 2 H2O}}
- {{#Chem: Ca(COO)2 { = ≥600°C } CaCO3 + CO|t=600}}
via nitric acid
- Combine urine and nitric acid, producing urea nitrate crystals
- Filter
- Discard filtrate
- Repeat
- Until no further residue is collected and the crystals are as pure as desired
Synthesis
industial
- Combine carbon dioxide with ammonia [1]
- {{#Chem: CO2 + 2NH3 = CO(NH2)2 + H2O}}
Wohler
- Combine silver cyanate with ammonium chloride.
- {{#Chem: AgNCO + NH4Cl → CO(NH2)2 + AgCl}}
See Also
References
- ↑ US patent 4061675A
Link courtesy Google